|
Developers:
|

|
Adele P. Hudson
Horatio B. Hackett School
Philadelphia, PA
|
E. J. Blusiewicz
Rohm and Haas Company
Bristol, PA
|
|
|
|
Grade
Levels:
|

|
4 and 5
|
|
|
|
Disciplines:
|

|
Physical Science
|
|
|
|
Goals:
|

|
- To identify chromatography as a
method of separating the components of a liquid.
- To develop an understanding that
dyes are composed of more than one component in achieving
a particular color.
- To become familiar with various
Chromatography techniques for separating these
components.
|
|
|
|
Background:
|

|
Chromatographic separation enables a
chemist to separate, isolate, quantify and identify the
components of a mixture.
Chromatography in the broadest sense
refers to processes that permit the resolution of a mixture
as a consequence of differences in rates at which the
individual components of that mixture migrate through a
stationary medium under the influence of a mobile
phase.
Paper Chromatography is a method of
separation in which a solvent passes through filter paper.
The mixture is introduced on the filter. The end of the
filter paper is placed in water and the capillary action of
the filter causes the solvent to flow. The components that
exhibit better solubility characteristics in the solvent
move at a faster rate. The result is the formation of moving
bands of color.
The glass column technique involves
packing a glass column with adsorbents such as silica,
alumina, or calcium carbonate. The sample is introduced at
the top of the column. Sufficient solvent is added until
each of the adsorbed components has been removed and
collected.
|
|
|
|
Teacher's
Note:
|

|
The principle aim of this week's work
in Project Labs was to give the teacher an opportunity to
work with an expert in the area of Industrial Chemistry. The
result of this experience was the preparation of new lessons
that are both motivating and scientifically correct. It is a
completely new approach to the teaching of science.
The result will provide the student
with an appreciation for the over-all analytical process. At
the same time developing basic laboratory techniques as well
as strengthening student's process skills.
Chromatography at first glance may
appear to be too difficult for the young student at the
elementary level. It is presented in such a manner that it
is viewed as only a part of the whole picture. It is a
representative sampling of some of the methods for the
completion of an analysis.
The needs of the student's
developmental stage are met and challenged. The progression
of such will move the student into the next stages with
assurance. The final result will be a student with firm
understanding of advanced scientific theory and the skills
for the operation of new technological equipment.
|
|
|
|
References:
|

|
"Modern Liquid Chromatography"; L. R. Snyder & J. J.
Kirkland; John Wiley & Sons, NY; 1974.
"Fundamentals of Analytical Chemistry"; Douglas A. Skoog
& Donald M. West; Holt Rinehart & Winston, NY; 1966.
List of Materials And Where To Buy Them
|
Fisher Scientific
1-800-635-9451
50 Valley Stream Parkway
Great Valley Corporate Center
Malvern, PA 19355
Whatman Filter Paper #1 Diameter 18.5
Whatman # 1001-185
Cat. # 09-805H 100/10.65
Cost $10.65
Wiretrol Calibrated Micropipets Cap 10
Orange
Cat # 21-175B 100/12.50
Cost $12.50
Short-Length Micro Caps Cap 10
Cat #21-170F
Cost $7.50
|
Silica Gel Adsorption 25g
Whatman # 4119-025
Cat #05-713-368
Cost $12.50
Medicine Droppers St. Tip
Cat #13-700 144/23.00
Cost $23.00
Thomas Scientific
(215) 988-0533
Micro Extension Clamp
Cat. #2826-C83
Cost $13.00
Sand Hardware Store
Vegetable Oil Super Market
Food Dyes Super Market
Flair Pens Stationers
|
|
|
|
|
Experiment
I:
|
|
Index of Experiments Showing
Logical Progression
Paper Chromatography To Demonstrate
Chromatography in General
|
|
Experiment
II:
|
|
A Comparison through Separation and Isolation Using Paper
and Column Chromatography.
|
|
Experiment
III:
|
|
A simplistic approach to show partitioning between two
liquids. To demonstrate what happens in paper and column
chromatography experiments where partitioning occurs between
a liquid and a solid.
|
|
|
|
Experiment
I:
|
|
Determining The Best Paper To Use For Separating Ink
Components
|
|
Objective:
|
|
- To determine which filter paper is best for
separating the components of black, green, blue and red
ink in Flair� Pens.
- Students will record colors observed in the
separation process.
- Students will measure and graph results.
|
|
Materials:
|
|
Different grades of filter paper or different brands of
paper towels
3 to 4 colors of Flair� Pens
3 to 4 100-mL jars with lids
Water bottle filled with tapwater.
|
|
Procedure:
|
|
- Cut strips of paper so that strips do not touch sides
of jar when inserted. The strips must be long enough to
overlap the rim of the jar.
- Fill 100-mL jars 1/8 full with tap water. Use a water
bottle to prevent sides of jar from getting wet.
- Place 3 dots of each colored ink 1.5 cm from bottom
of testing strip. Use a separate strip for each
color.
Caution, the dots must be the smallest that you can make
them.
- Carefully immerse only the lower part of strip in
water.
- Cap the jar being careful not to create turbulence in
the water.
- Allow to develop until the water front moves past the
original ink dots by about 10 cm.
- Record the color changes.
- Carefully remove strips and allow to dry. Mount on
sheet of paper.
- Measure and record lengths of color bands
|
|
Data:
|
|
Color Bands
|
|
Paper #1
|
Paper #2
|
Paper #3
|
Paper #4
|
|
Black
|
|
|
|
|
|
Green
|
|
|
|
|
|
Blue
|
|
|
|
|
|
Red
|
|
|
|
|
|
|
Experiment
I:
|
|
Lengths of Color Bands
Black______________________________________
Green _____________________________________
Blue ______________________________________
Red _______________________________________
Construct a Bargraph of the colors of each ink.

|
|
Reviews:
|
|
|
1.
|
Which paper had the least streaming of the color
band?
|
|
2.
|
Which paper had the most streaming of the color
bands?
|
|
3.
|
Which paper showed the best color separations?
|
|
4.
|
Which paper would you use to do other tests of
this kind?
|
Extensions
- Repeat the experiment but substitute different pens.
- Record results in the same manner.
- Finally, repeat the experiment by selecting two of
the same color but different manufacturers. Make a
comparison and record results.
|
|
|
|
Experiment IIa
|
|
A Comparison of Paper Chromatography and Column
Chromatography
|
|
Materials:
|
|
Paper strips (towels or filter paper) cut to fit jar not
touching sides and overlapping rim of jar.
100-mL glass jars with lids
Blue, green, yellow and red food dye
Micro caps
|
|
Procedures:
|
|
- Fill 4 (100-mL) jars 1/8 full with tap water. Use a
water bottle to prevent sides of jar from getting wet.
- Place three dots of dye 1.5 cm from bottom of paper
strip. Use a microliter cap to make dots. Use a separate
strip for each color.
- Carefully immerse only the lower part of strip in the
water. Do not allow the water to touch the dots.
- Cap the jar being careful not to create turbulence in
the water.
- Allow to develop until the water front moves past the
original ink dots by about 10 cm.
- Record the color changes
- Allow to dry and mount on sheet of paper.
|
|
Data:
|
|
Food Dye Color Bands
Black______________________________________
Green _____________________________________
Blue ______________________________________
Red _______________________________________
Tape Dye Paper Samples
|
|
Experiment IIa
|
|
A Comparison of Paper Chromatography and Column
Chromatography
|
|
Objective:
|
|
To separate food dye into its component parts by using a
liquid chromatography column and to compare this with
results from paper chromatography experiment.
|
|
Materials:
|
|
|
8cm rubber bulb pipettes
|
Washed sand
|
|
Glass wool
|
Blue and green food dye
|
|
Silica gel
|
|
|
|
Procedures:
|
|
- Remove the rubber bulb from the glass pipette.
- Pack a small amount of glass wool in the narrow end
of the pipette. Add a layer of silica gel, approximately
3 cm. Add a 3mm layer of washed sand to the top of the
silica gel. Carefully drop 10 microliters of blue dye to
the top of the sand. Add 2 drops of water to start the
dye through the sand. After the water has passed through
add 2 more drops. Wait until this has passed through the
sand. At this point fill the tube with water.
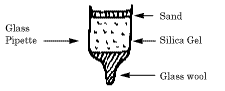
- Collect the liquid from the separated layers.
- Continue to add water in small amounts until all of
the bands of color have been washed out.
- Repeat procedure with green dye.
- Save the collected material in separate bottles.
|
|
Discussion:
|
|
While the two methods, paper chromatography and column
chromatography give similar information, there are
differences. What are the differences and when would you use
each method or possibly both methods?
|
|
Teacher's
Note:
|
|
Paper chromatography is used primarily to determine if a
material is pure or a mixture and in some cases can be used
to identify the presence of a material by comparing the
distance an unknown travels as related to how far a known
travels. Column chromatography is usually used to separate
and isolate a quantity of a material from a mixture, to
purify a material, i.e., separate it from impurities, and to
separate and identify components of a mixture. Both methods
are used when paper chromatography is used to determine if a
separation is possible and what solvents are best to achieve
that separation.
|
|
|
|
Experiment III
|
|
Partitioning in Water and Oil
|
|
Objective:
|
|
To demonstrate partitioning of materials in an
aqueous or oil base.
|
|
Materials:
|
|
|
Light colored olive oil
|
Fresh fruit dye
|
|
Water
|
Kerosene soluble dye
|
|
Commercially prepared food dyes
|
Pollen
|
|
|
Procedures:
|
|
- Use 2 small jars with lids. Select 2 colors of food
dye. Fill each jar 1/4 full with water. Pour an equal
amount of oil to the water. Add 2 drops of food dye. Put
the lid on the jar and shake. Observe and record the
results.
- Repeat step 1 but for the sample add 6 drops of
colored kerosene. Observe and record results.
- Repeat step 1 but for the sample add 2 drops of fruit
dye. Observe and record results.
- Fill small jar with lid half full with water. Add the
pollen of a lily. Cap the bottle and shake. Observe and
record results. Repeat the process using oil.
|
|
Data:
|
|
|
Food Dye
|
Kerosene Dye
|
Blackberry
Strawberry
|
Pollen
|
|
Water soluble
|
Water insoluble
|
Water insoluble
|
Water insoluble
|
|
Oil insoluble
|
Oil insoluble
|
Oil insoluble
|
Oil insoluble
|
|
|
Conclusion:
|
|
Pollen displayed a solubility in both
water and oil. The color red was extracted in the water. The
color orange was extracted in the oil.
The food dye was soluble only in
water. The kerosene dye was soluble only in oil.
We wanted to demonstrate that
materials soluble in both phases would position between the
two phases. We were not able to find a "Safe" dye for the
purpose.
|



