Surface tension is a property of the surface of a liquid that allows it to resist an external force.
Surface tension is dependent on temperature. For that reason, when a
value is given for the surface tension of an interface, temperature must
be explicitly stated. The general trend is that surface tension
decreases with the increase of temperature, reaching a value of 0 at the
critical temperature. For further details see the Eötvös rule below. There are only empirical equations to relate surface tension and temperature:
- Eötvös:
Here V is the molar volume of that substance, TC is the critical temperature and k is a constant valid for almost all substances. A typical value is k = 2.1 x 10−7 [J K−1 mol-2/3]. For water one can further use V = 18 ml/mol and TC = 374°C.
A variant on Eötvös is described by Ramay and Shields:
where the temperature offset of 6 kelvins provides the formula with a better fit to reality at lower temperatures.
 is a constant for each liquid and n is an empirical factor, whose value
is 11/9 for organic liquids. This equation was also proposed by van der Waals, who further proposed that
is a constant for each liquid and n is an empirical factor, whose value
is 11/9 for organic liquids. This equation was also proposed by van der Waals, who further proposed that  could be given by the expression,
could be given by the expression,  , where
, where  is a universal constant for all liquids, and
is a universal constant for all liquids, and 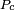 is the critical pressure of the liquid (although later experiments found
is the critical pressure of the liquid (although later experiments found  to vary to some degree from one liquid to another).
to vary to some degree from one liquid to another).
Both Guggenheim-Katayama and Eötvös take into account the fact that
surface tension reaches 0 at the critical temperature, whereas Ramay and
Shields fails to match reality at this endpoint.
The Eötvös Rule
The Eötvös rule, named after the Hungarian physicist Loránd (Roland) Eötvös (1848–1919) enables the prediction of the surface tension of an arbitrary liquid pure substance at all temperatures. The density, molar mass and the critical temperature of the liquid have to be known. At the critical point the surface tension is zero.
The first assumption of the Eötvös rule is:
1. The surface tension is a linear function of the temperature.
This assumption is approximately fulfilled for most known liquids. When plotting the surface tension versus the temperature a fairly straight line can be seen which has a surface tension of zero at the critical temperature.
The Eötvös rule also gives a relation of the surface tension behaviour of different liquids in respect to each other:
2. The temperature dependence of the surface tension can be plotted for all liquids in a way that the data collapses to a single master curve. To do so either the molar mass, the density, or the molar volume of the corresponding liquid has to be known.
For more information:
https://en.wikipedia.org/wiki/Surface_tension#Influence_of_temperature
https://en.wikipedia.org/wiki/E%C3%B6tv%C3%B6s_rule
Source: Wikipedia (All text is available under the terms of the GNU Free Documentation License and Creative Commons Attribution-ShareAlike License.)



